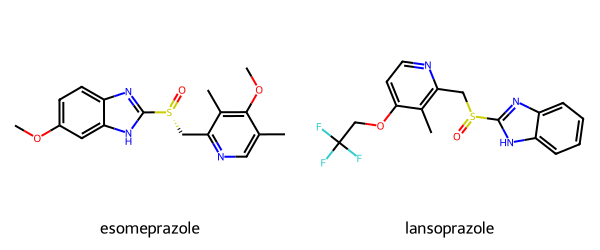
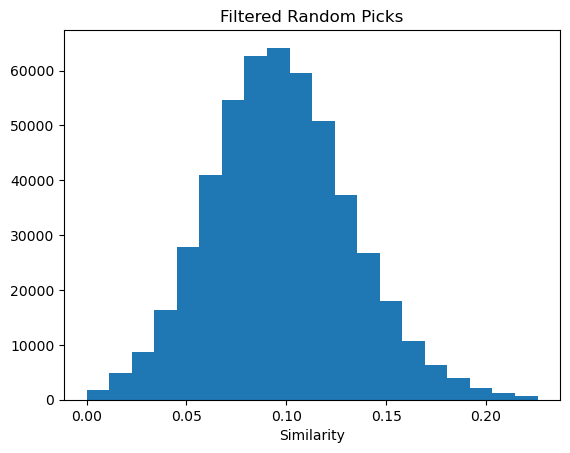
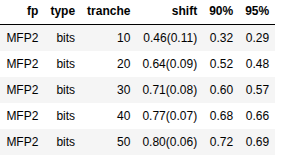
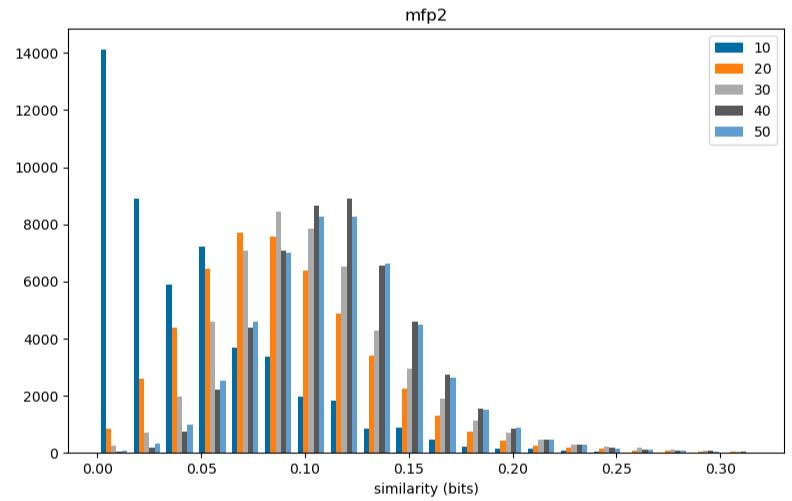
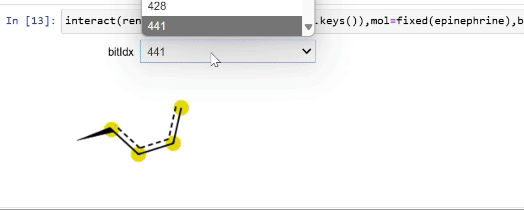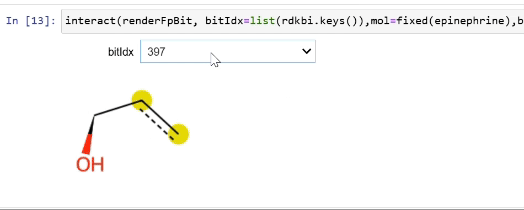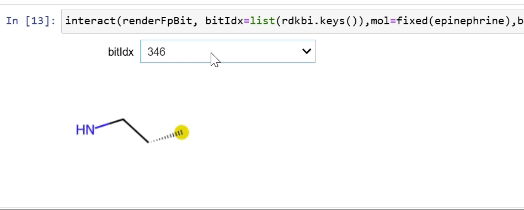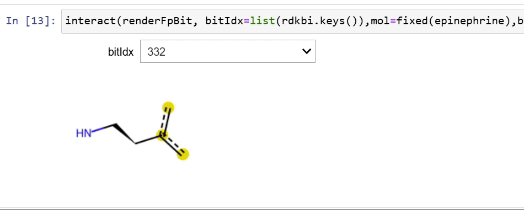
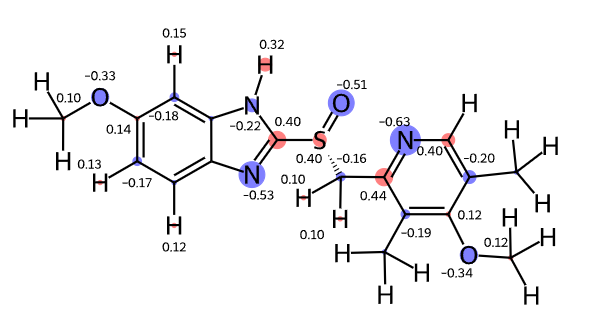
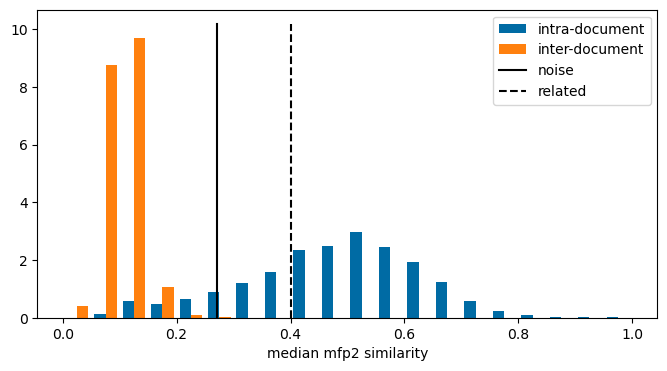
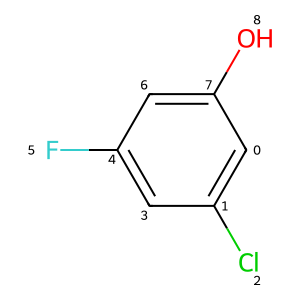
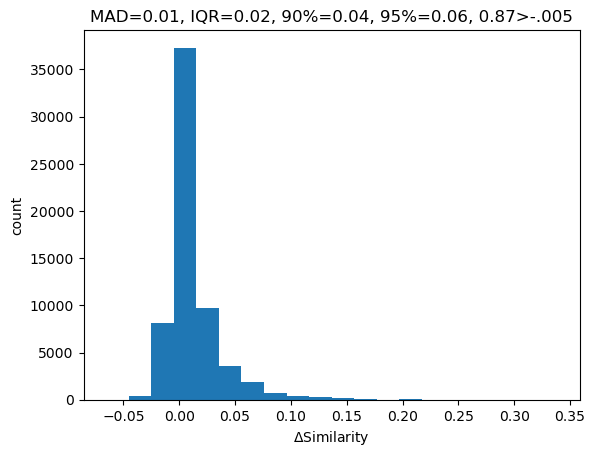
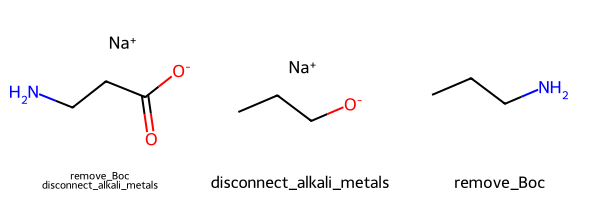
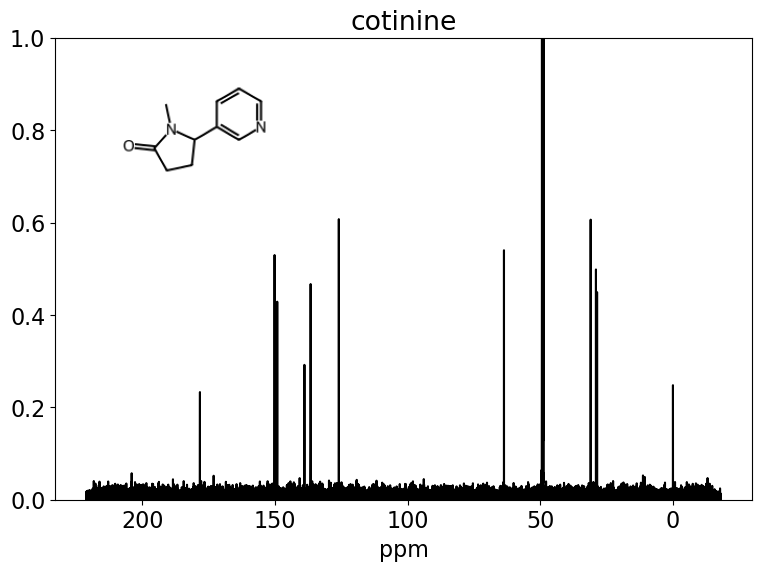
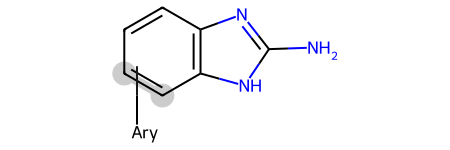
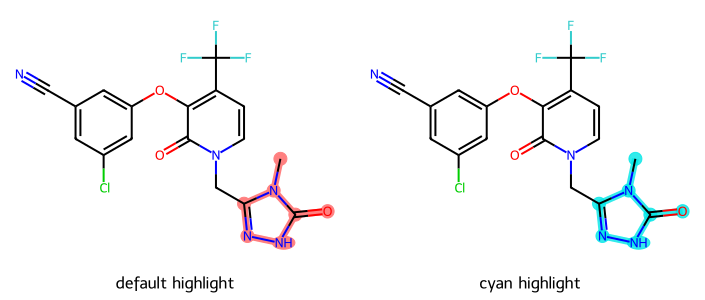
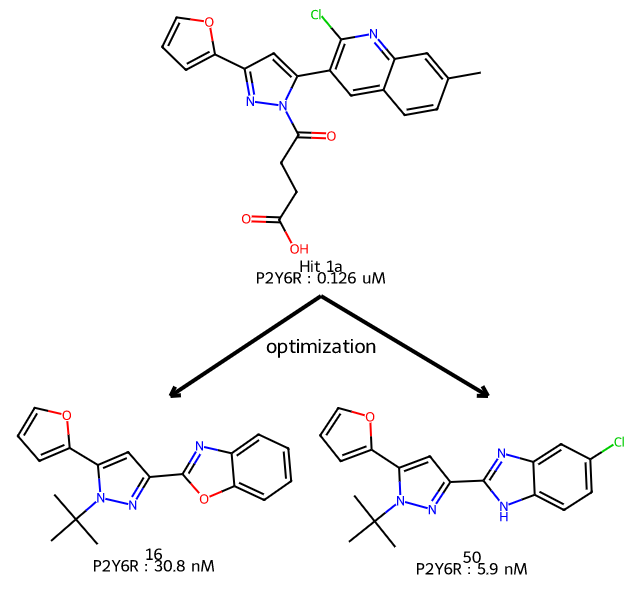
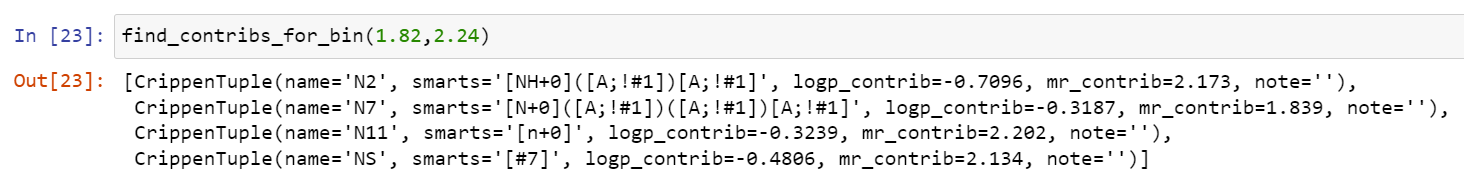
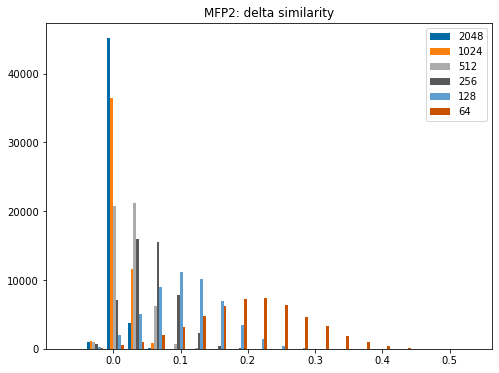
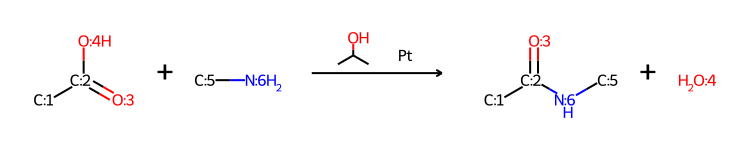
Please stop saying “The Tanimoto similarity is”
similarity
fingerprints
rants
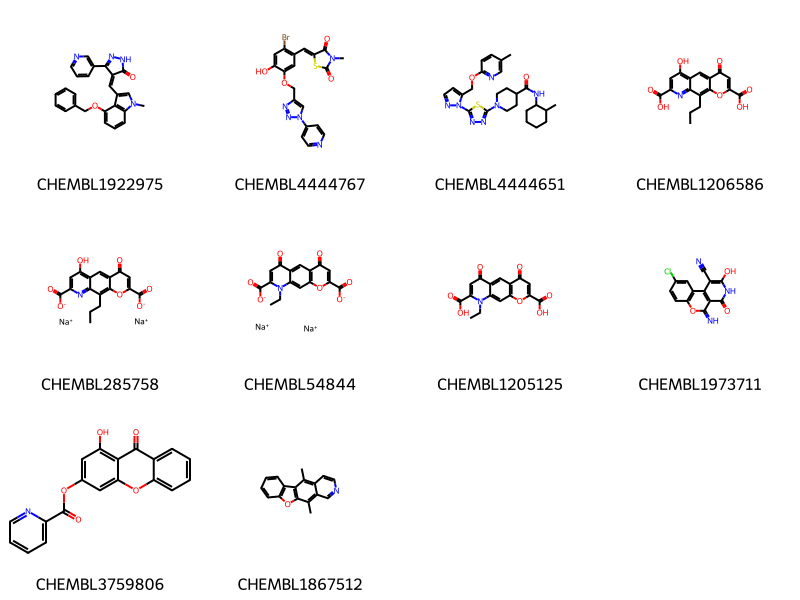
Picking a small set of diverse molecules from a large set
similarity
fingerprints
tutorial
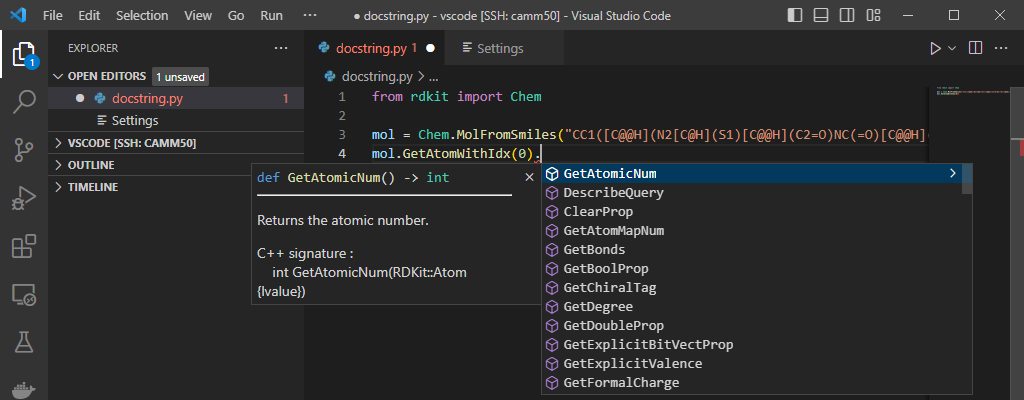
Sanitization options and molecule parsing.
documentation
technical
The impact of molecular size on similarity.
reference
similarity
fingerprints
Using custom fingerprints in PostgreSQL
cartridge
fingerprints
tutorial
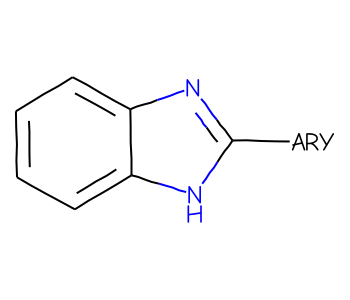
Explaining enhanced stereochemistry
prototypes
tutorial
stereochemistry
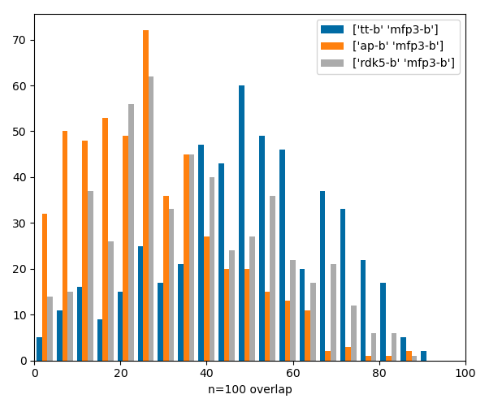
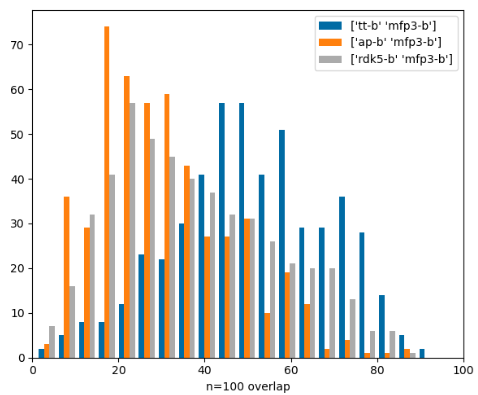
Similarity-search hitlist overlap part 2
reference
similarity
fingerprints
Rendering fingerprint bits
tutorial
drawing
fingerprints
Similarity-search hitlist overlap
reference
similarity
fingerprints
Building a similarity comparison set
reference
similarity
Tuning substructure queries
tutorial
substructure
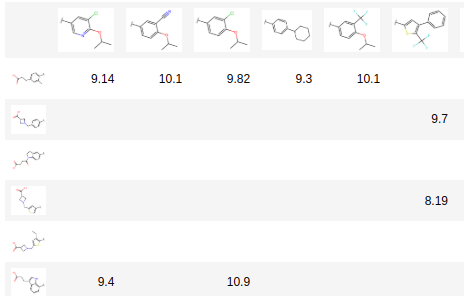
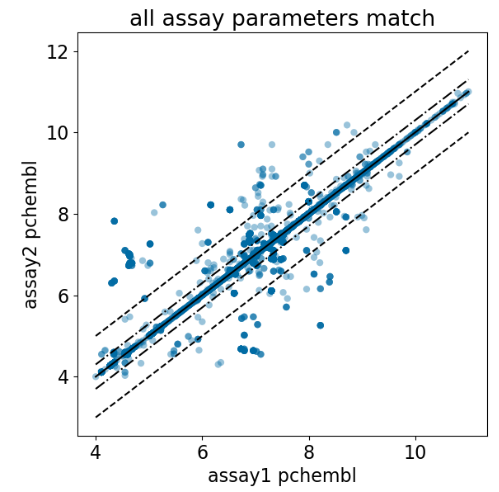
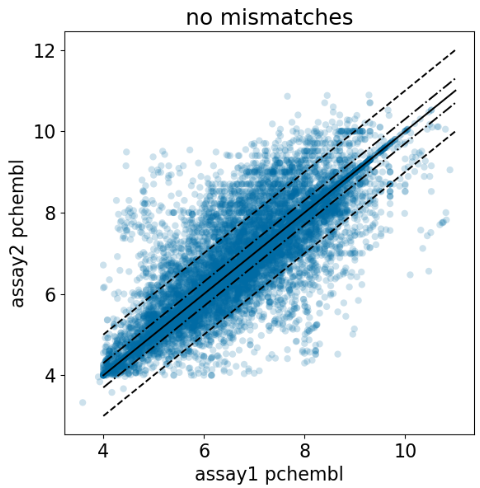
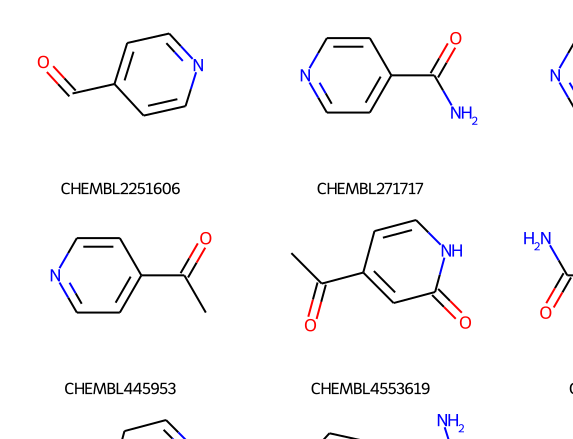
ChEMBL Document Similarity
exploration
similarity
Using the ACS1996 drawing style in PandasTools
documentation
tutorial
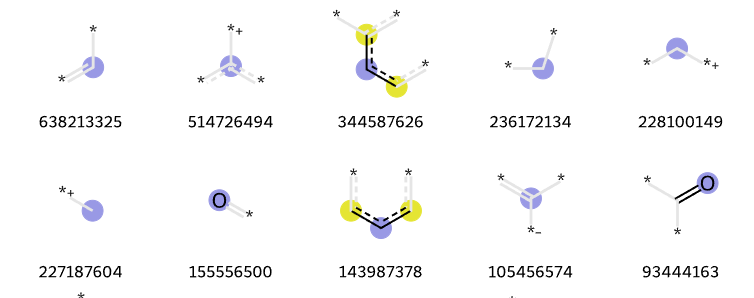
Additive fingerprints
exploration
similarity
fingerprints
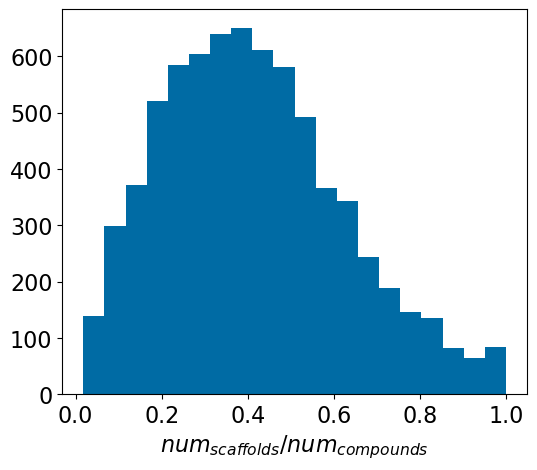
The problem(s) with scaffold splits, part 1
machine learning
rants
reference
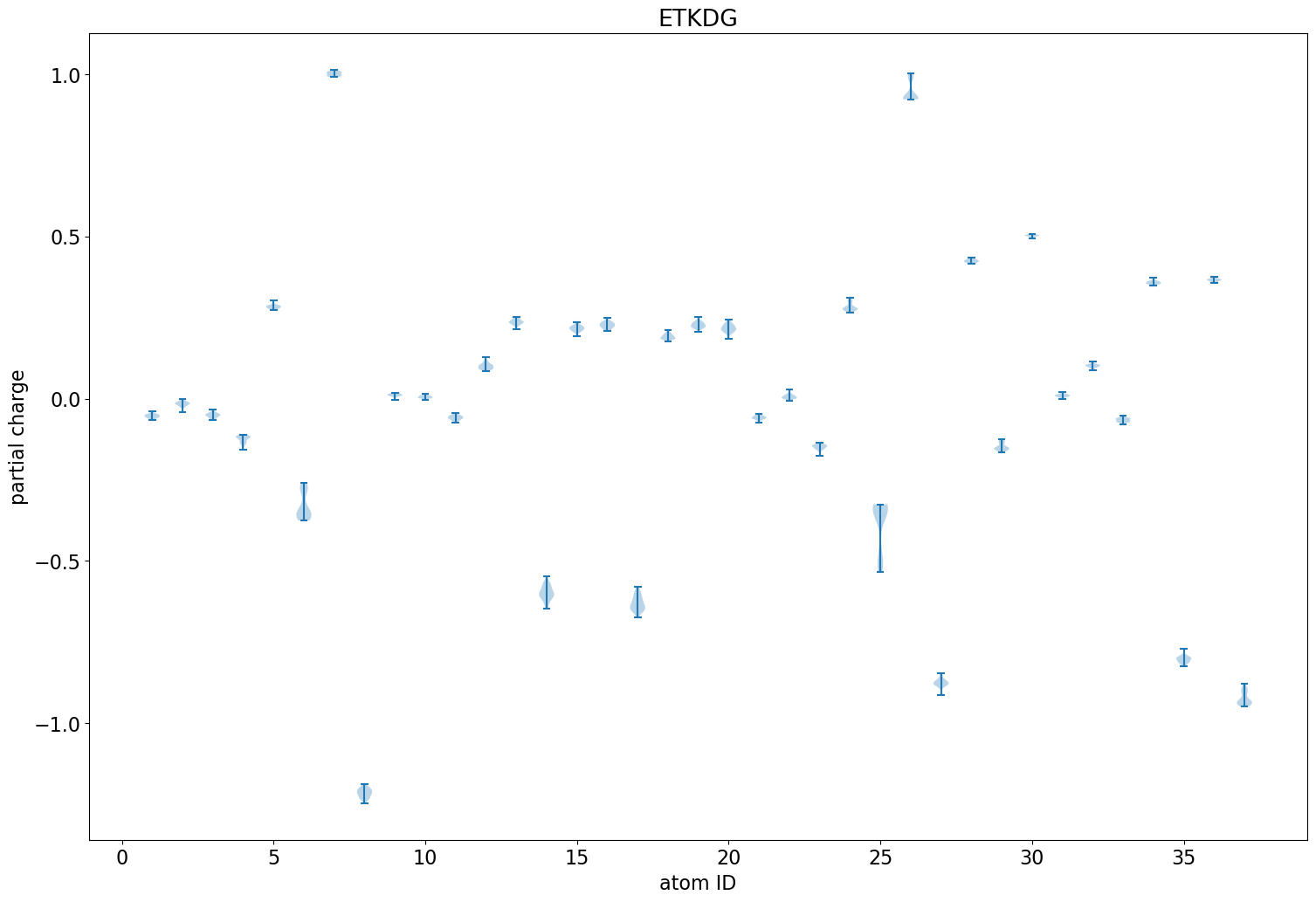
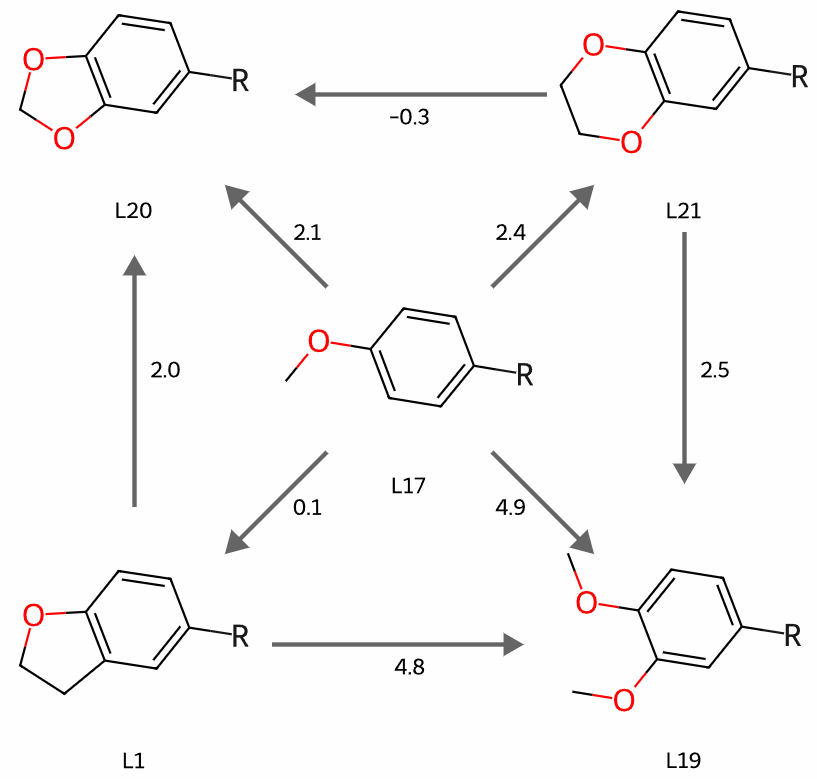
The impact of single-atom changes on similarity
fingerprints
reference
exploration
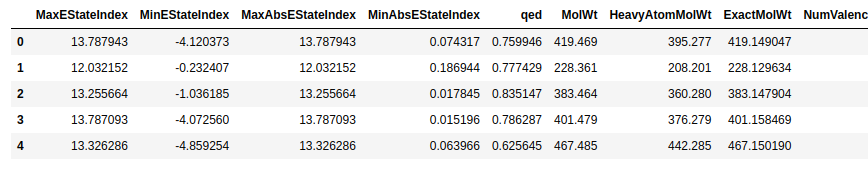
Calculating the SA_Score and NP_Score descriptors
tutorial
documentation
descriptors
Intro to Stereo Groups and Enhanced Stereochemistry
tutorial
stereochemistry
documentation
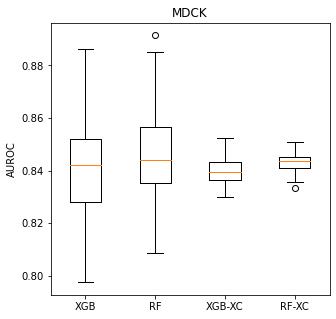
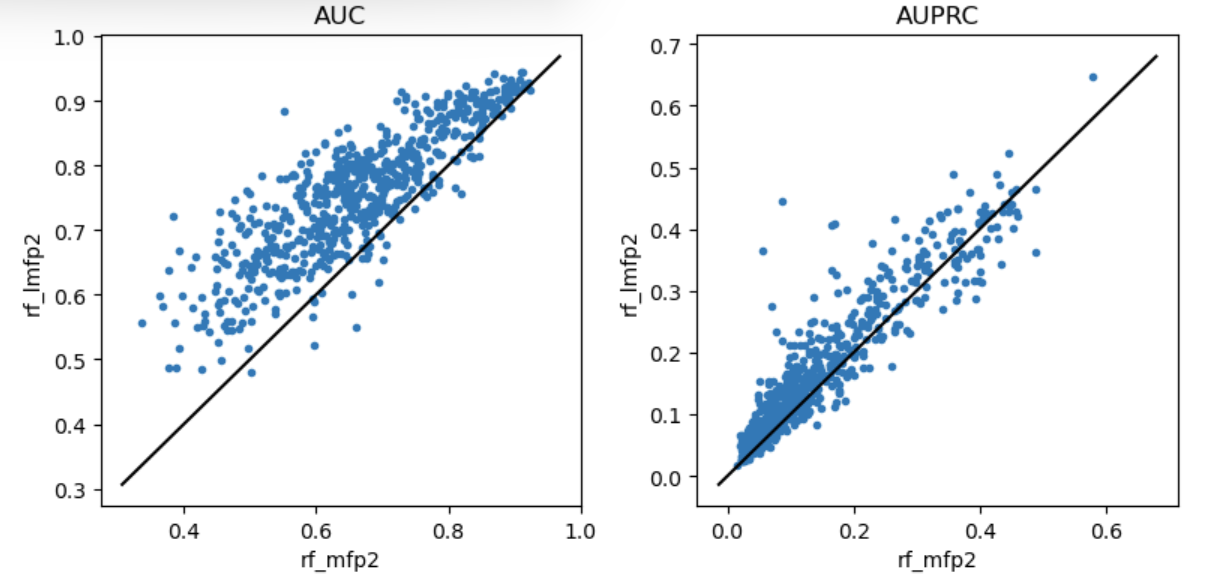
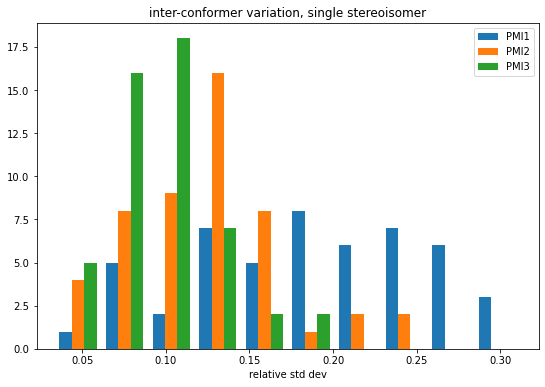
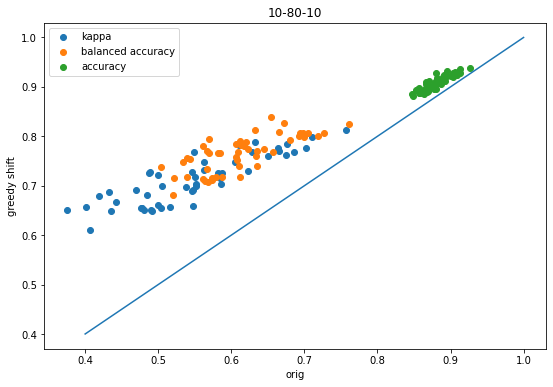
Variability of x-fold cross validation results
machine learning
exploration
Binary molecules and the cartridge
cartridge
tutorial
exploration
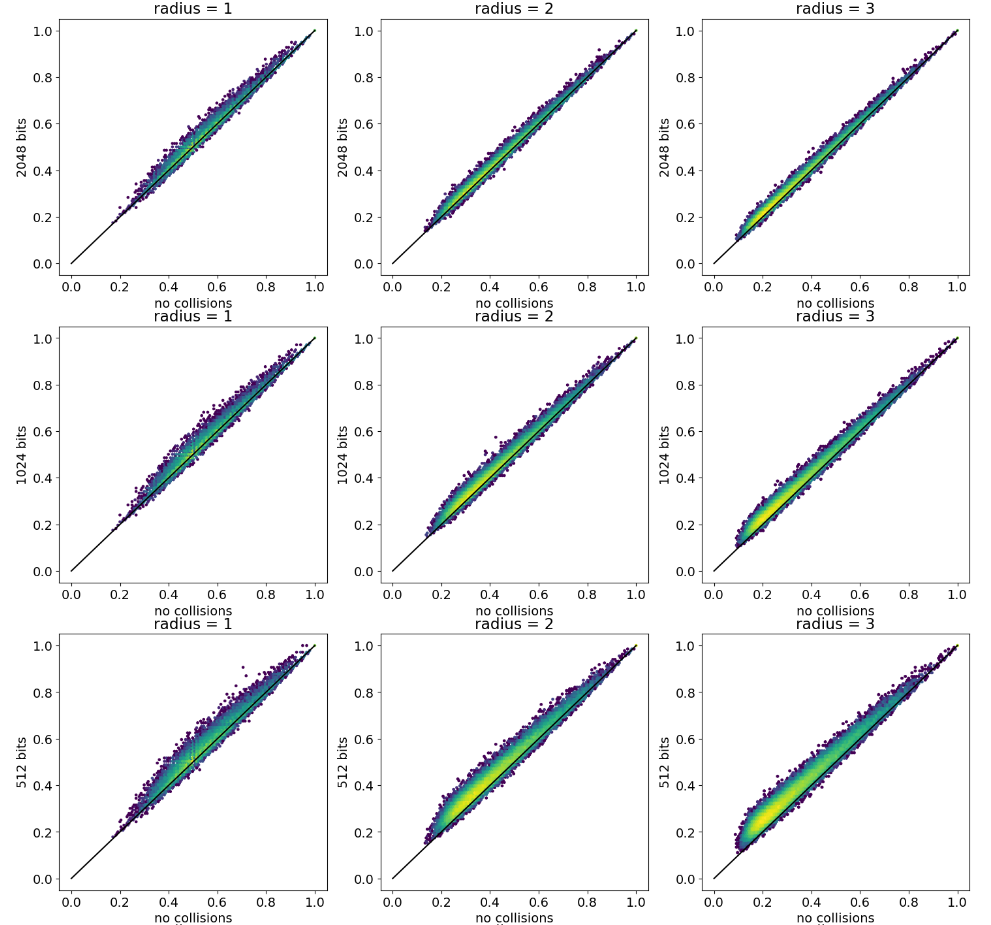
Colliding bits III, expanded
reference
fingerprints
Colliding bits II, revisited
reference
fingerprints
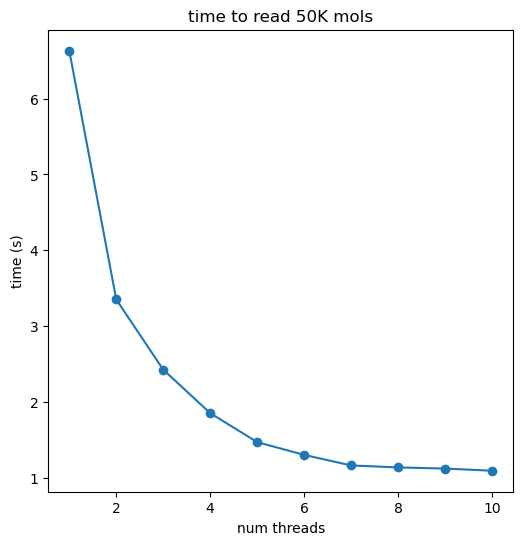
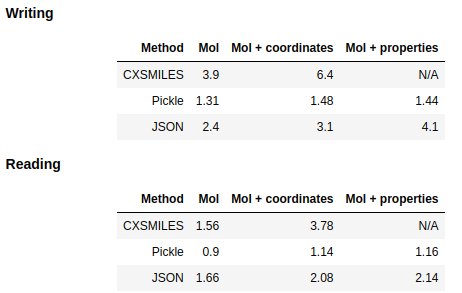
Timing methods for serializing molecules
reference
optimization
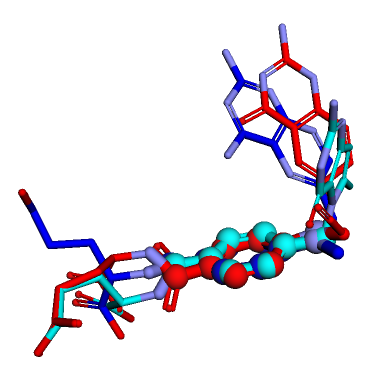
Dealing with multiconformer SD files
3d
conformers
tutorial
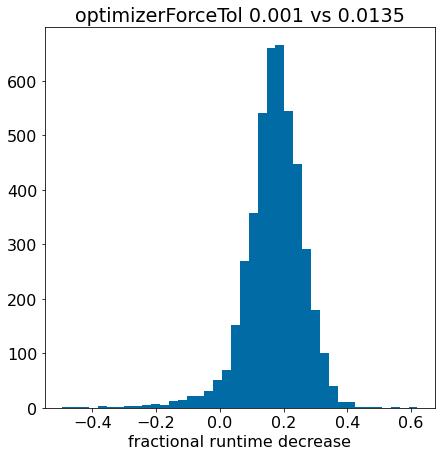
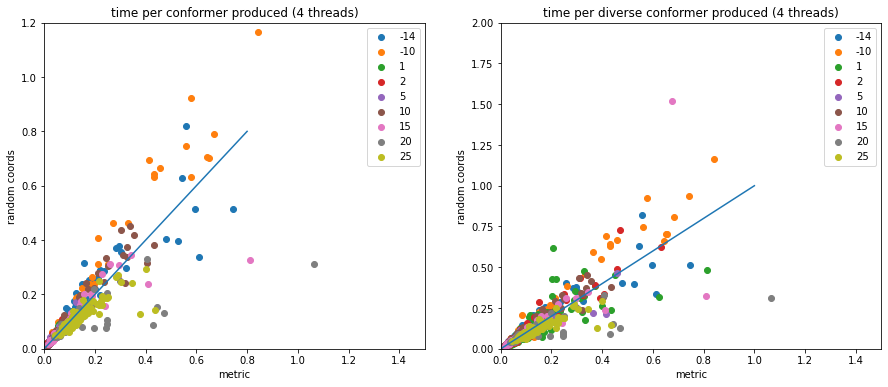
Optimizing conformer generation parameters
3d
conformers
optimization
A Ternary GHOST
exploratory
machinelearning
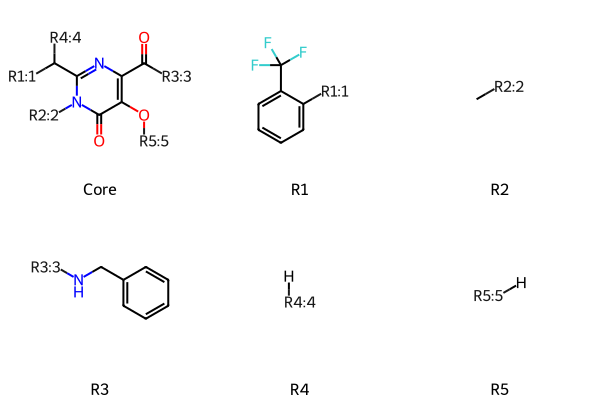
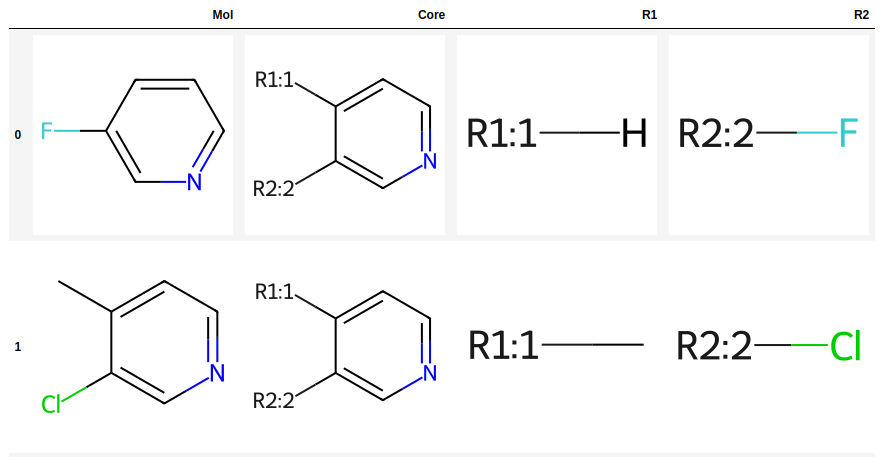
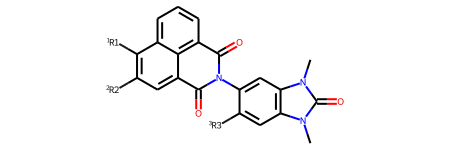
R-Group Decomposition and Highlighting
tutorial
prototypes
drawing
rgd
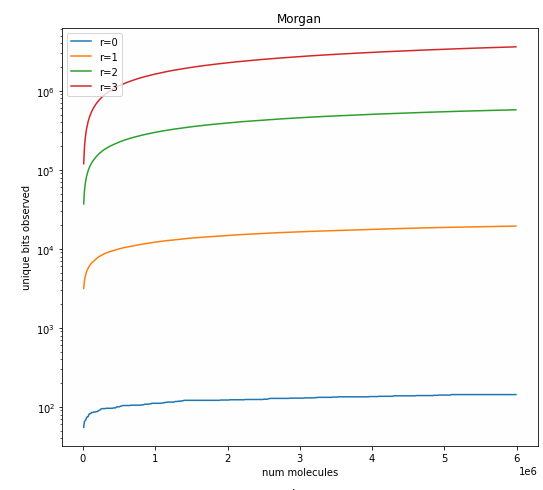
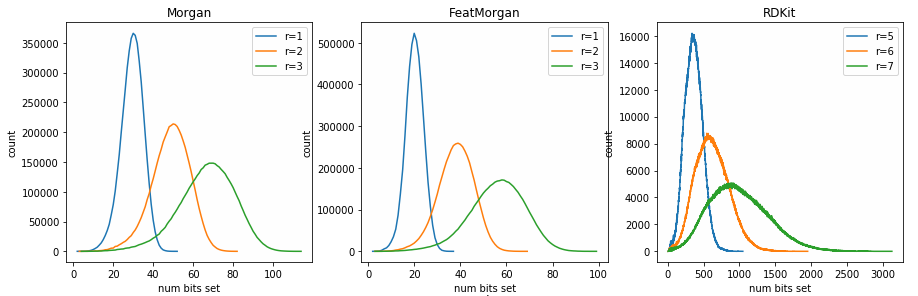
Looking at the number of bits set by different fingerprints
fingerprints
reference
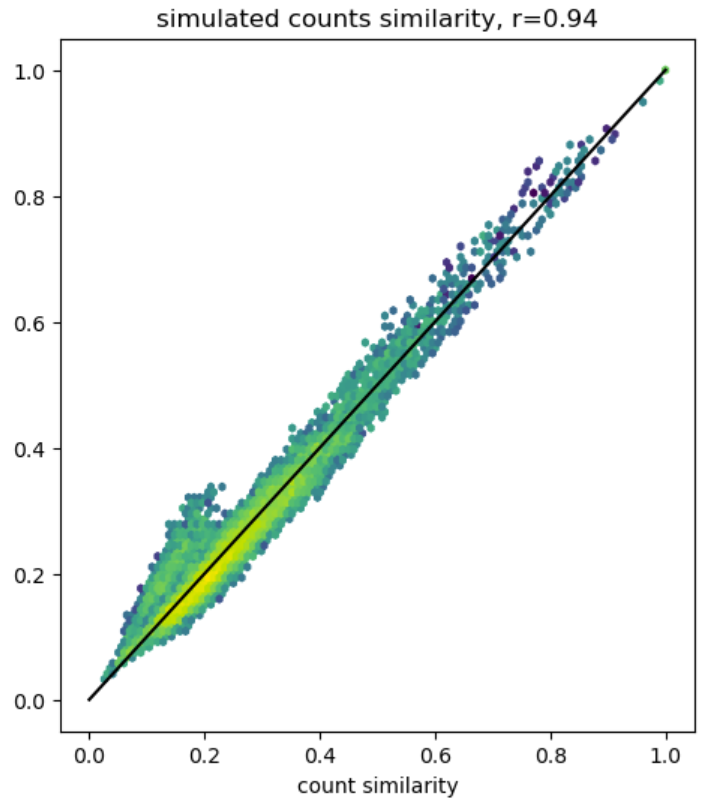
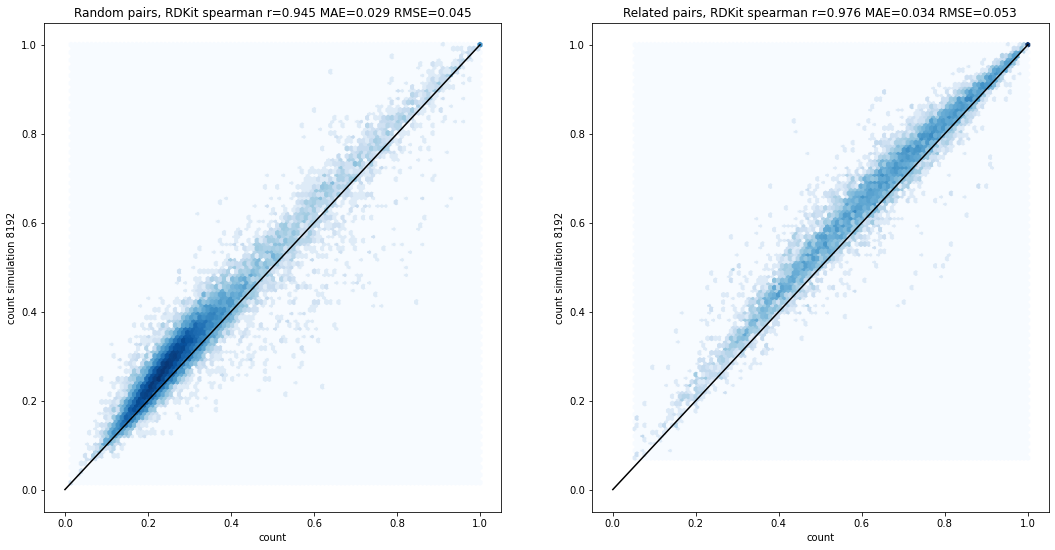
Simulating count fingerprints
fingerprints
technical
reference
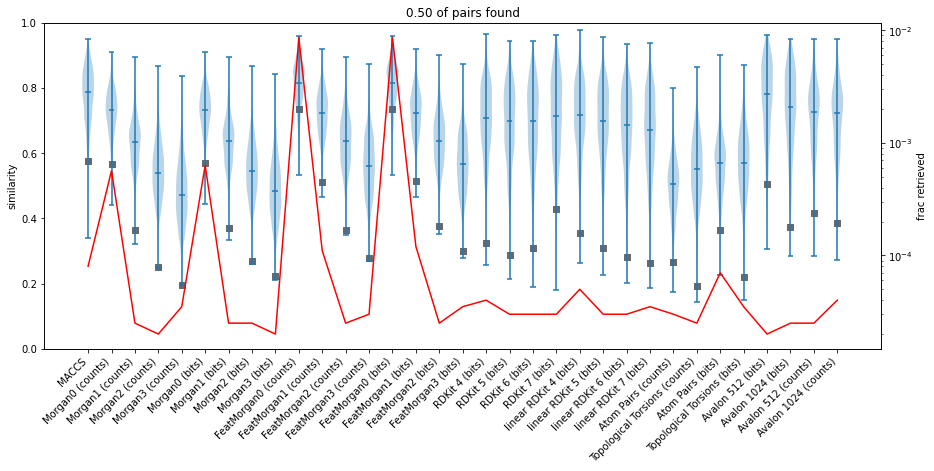
Fingerprint similarity thresholds for database searches
similarity
reference
Thresholds for “random” in fingerprints the RDKit supports
fingerprints
similarity
reference
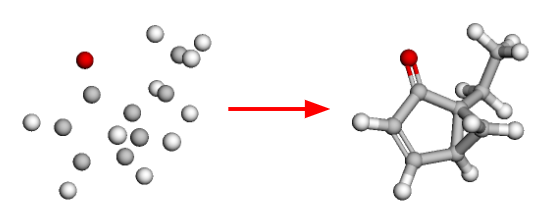
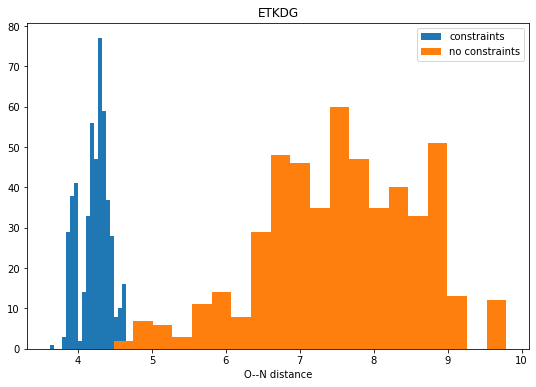
Looking at random-coordinate embedding
conformers
exploration
3d
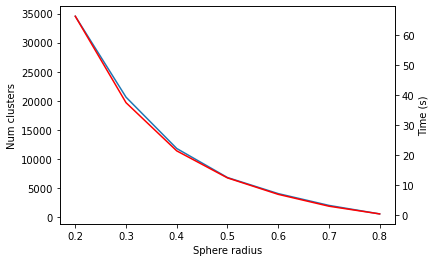
Sphere exclusion clustering with the RDKit
similarity
tutorial
No matching items